Stress Corrosion Cracking (SCC) is an environmentally assisted failure caused by exposure to a corrodent while under a sustained tensile stress. SCC is most often rapid, unpredictable and catastrophic. Failure can occur in as little as a few hours or take years to happen and is commonly observed in the absence of other forms of corrosion, such as general or crevice corrosion.
Most alloys are susceptible to SCC in one or more environments requiring careful consideration of alloy type in component design. In oxygenated aqueous chloride environments austenitic stainless steels and many nickel based alloys are known to perform poorly. These alloys demonstrate a rapid decrease in failure time with increasing applied stress. Use of these alloys often requires additional processing, such as shot peening, cathodic protection or the application of protective coatings to prolong the structure life. However, the additional processing requirements add significantly to the cost of use.
Numerous service conditions exist where high chloride conditions are present. Aqueous chloride environments are common in the Oil and Gas industry in both the downhole and production situations. Highly concentrated hot brine or potassium chloride are common in exploration drilling requiring the use of chloride SCC resistant materials such as high copper alloys. Seawater conditions are another area where copper beryllium alloys, for example, have been exceptionally successful for undersea electronic and fiber optic housings.
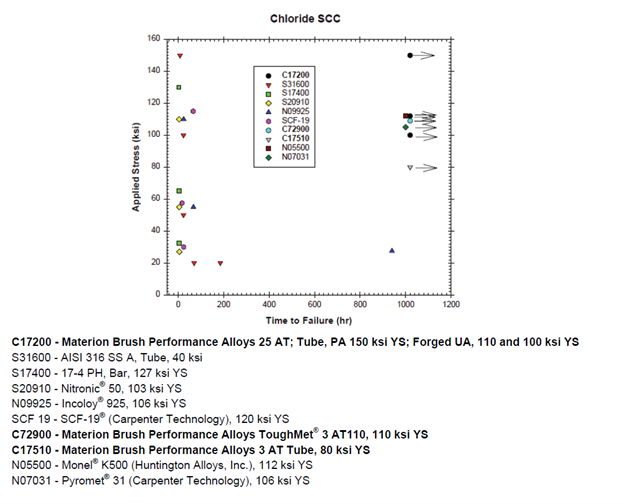
A method of commonly evaluating material susceptibility to Chloride SCC is ASTM Standard Practice G36, “Performing Stress-Corrosion Cracking Tests in a Boiling Magnesium Chloride Solution”. This accelerated test method incorporates C-ring specimens of various alloys of steel, nickel and copper to determine the failure time as a function of applied stress. The specimens evaluated were loaded to an applied stress of 100% of the yield strength and immersed into the solution as described by the standard practice. If the specimens failed in less than 1000 hr, additional specimens were loaded to 50% of the yield strength and tested in solution. Copper beryllium alloy C17200 was tested at three levels of strength to determine the effect of aging on resistance to chloride SCC.
The results shown in the graph indicate that the stainless steel alloys are highly susceptible to chloride SCC, as are some of the nickel-based alloys. These alloys fail by cracking in times as short as several hours, even at a fraction of their yield strength. The data also show that several high performance copper-based alloys demonstrate SCC resistance at strength levels equal to, or greater, than several SCC resistant nickel alloys. Copper beryllium C17200, for example, provides the greatest strength and SCC resistance of the alloys evaluated. The excellent corrosion resistance is independent of strength, shown at 0.2% yield strengths ranging from 100 ksi to 150 ksi (689 MPa to 1034 MPa). A lower strength, but higher conductivity copper beryllium alloy, C17500, is also shown to be resistant to chloride SCC. A new commercially available spinodally hardened Cu-Ni-Sn alloy, ToughMet® 3 AT, offers SCC resistance at a yield strength of 110 ksi (758 MPa).
When design considerations require a chloride SCC resistant alloy, other material properties and attributes should also be considered. For example, AT0024/0800 instrumentation for directional sensing during downhole oil and gas exploration may require high strength and nonmagnetic behavior. Other downhole drilling situations require bending of a drill string over a tight radius defining the need for a low modulus alloy which reduces generated stress levels. Wear and friction behavior may be necessary for bushings and wear surfaces exposed to SCC inducing environments. These design requirements eliminate the high nickel alloys from the application because of their ferromagnetic behavior, high elastic modulus and propensity for galling and seizing. High strength copper alloys, such as the copper beryllium and spinodal alloys, are the engineering solution in many applications where the material demands are high.
Read our whitepaper on stress testing in a NACE environment.
Thanks for joining me for another edition of In Our Element. For ongoing industry updates, connect with us on LinkedIn.