One of the first things a coating operator asks when they are introduced to new coating materials to be used in the coating chamber (especially a multi-pocket e-beam chamber) is “how will the form of this material influence the process?” This is in fact an excellent question. Depending on the material (metal, opaque compound or glass/crystalline material), there are many shapes and sizes to choose from even within the same chemical family. These may include compound powders, granules, pieces, cones, or solid charges. While the most obvious answer is it affects cost and density, it is more complex than that. The manufacturing process - as well as the material - will impact the outcome.
Let us start with a “truncated cone” the most familiar pocket and workhorse holder for classical electron beam platforms. While other options do exist, choosing this as a starting point will set the foundation for more detailed discussion. Look at Figure 1(left) and imagine filling the cone with marbles. Then view the chart of reported1 random filling packing density and the aspect ratio (defined as height over the base diameter of a simple cone) to find an optimum.
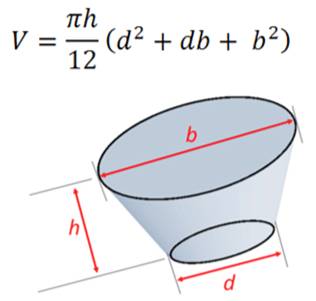
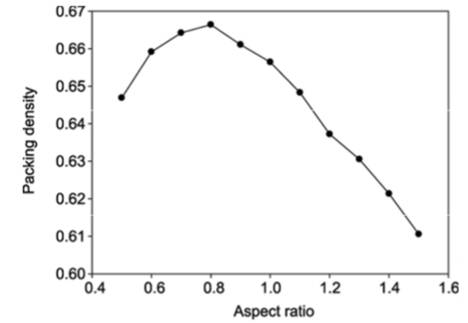
Figure 1: Truncated Cone Volume & Optimum Packing in Truncated Cone1
Note that as the volume shrinks, or the ball size increases, the fewer balls (less coating material at that size) will fill that space. Also, the larger the gaps will be at interfaces with other balls and the walls of the crucible. In the practical world, the balls are tablets, shot or uniform granules which must provide enough material on hand for the coating run while minimizing the space between the adjoining particles.
To mitigate spit and particle explosions, the surface area of the charge must be minimized and the density of the particle customized to the deposition energy. A bigger pocket or a maximum aspect ratio (pan or trough) will influence the complexity of the burn down and use (stability) of the coating process.
When answering the call for higher packing density and reduced spit with dielectric compounds, we must balance the yield associated with a pre-consolidated powder or melted ingot - to the mixed distribution of granules. (We will leave metal shot, slugs and wire for another discussion). While generally more expensive, higher-density melted or pressed powders have several benefits. They are less prone to producing dust, or exiting the pocket or tunneling during deposition processes. Further protection comes from manipulating particle density, phase purity and size distribution. Custom size distributions can increase the packing density > 75% and maximize the utility for classical crucibles from 15cc to 40cc.
Before addressing true premelts, it is important to introduce the limitations of handling crystalline materials that melt and cool. These limitations apply to non-glassy oxides and the crystalline fluorides critical for optical coating.
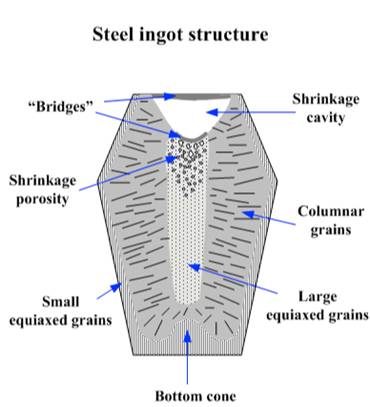
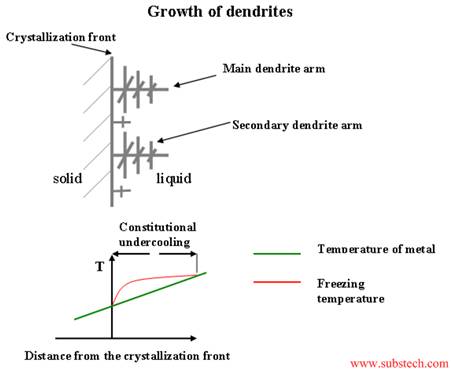
Figure 2: Classic Structures during Cooling of a Polycrystalline System
For illustration purposes, it is easier to use steel ingots in the Figure 2 graphics. A key concept is that steel, just like magnesium fluoride and titanium dioxide, has good and bad characteristics which influence the yield (cost) and usability. As solidification proceeds from the cooler skin of the vessel, small changes in composition and the temperature gradient accelerate dendrite release i
It becomes even more complicated as the solidification process occurs repeatedly in the pocket each time the material is used. Visualize the image in Figure 2 being duplicated at each tablet or large melted area. For example, in the case of titanium dioxide, the starting material selection and refill conditioning is key to controlling the formation of detrimental phases like Ti2O3, Ti4O7. Other phases like these may be more prone to explosion and disrupting coating stability.
We employ multiple technologies to improve coating materials and processes. Among them is production of materials in tablet forms that are difficult to melt due to their hardness or stablility (GdF3 and HfO2); optimized powder consolidation; and advanced manufacturing technology for materials that are more challenging to melt (Fused SiO2, AlF3, ZnS).
We have also invested in key melting technology for crystalline systems that are challenged by purity, stress and sub-phases (MgF2, LiF, Ti3O5) and developed new casting and melting technology for intense photonics and semiconductor components (EB HfO2, EB Al2O3, EB Y2O3). We are continuing to navigate the complex issues with 99-100% dense starter charges as certain metals and some IR fluorides require the most homogeneous starter charges to meet challenging applications.
We take great pride in understanding the needs and challenges of our coatings customers and delivering quality products and services for their success in the thin film industry.
1. Li ShuiXiang, Chinese Science Bulletin January 2010 “Maximum Packing Densities of 3D Objects,” P. 116 “Cone”, Figure 2 and Definition of Aspect Ratio with Result.